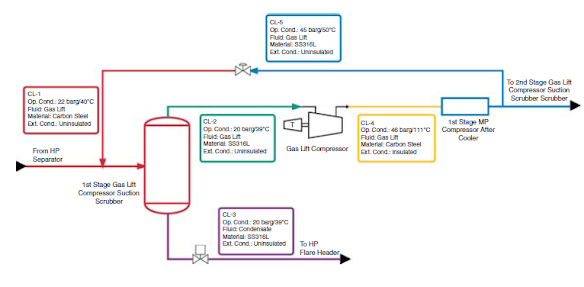
Corrosion loop is used to simplify inspection procedures in refining processes.
Corrosion loop is defined as a group of components with common materials, processes, and operating parameters.
It is needed when the scenario has -
1. Complex process
2. Several variables such a materials of construction, process parameters, functions
3. Multiple damage mechanisms


Comments
Post a Comment
Welcome to the CORROSPECTIVE BLOG! Scroll along for more posts! Looking forward to your feedback!
P.S. Don't forget to check out the free quizzes!